Caffeine Addiction or Chemical Overload – Could Decaf be More Chemically Dangerous than Caffeine?
 Mmmmmm….Decaf. Same great taste, same great chemicals? For a long time, I’ve drank decaf beverages without really giving a second thought to how my drink came to be “decaffeinated.” They grow decaf tea leaves and coffee beans, right? Right?
Mmmmmm….Decaf. Same great taste, same great chemicals? For a long time, I’ve drank decaf beverages without really giving a second thought to how my drink came to be “decaffeinated.” They grow decaf tea leaves and coffee beans, right? Right?
Not quite. Benzene, methylene chloride (dichloromethane), trichloroethylene, and ethyl acetate are all chemicals that have been or are used in the decaffeination process of tea, coffee, and kola nuts (those things that make coca cola taste so amazing!). Somehow, knowing their names makes that previously delicious cup of decaf suddenly sound a little more like a brew of hazardous toxins.
I’m an avid drinker of tea, and although my favorite saying is “While there is tea, there is hope,” I am also an avid believer of “8 hours of sleep a night will keep the crankiness out of sight!” So despite the many sneers of “What’s the point of decaf?” I keep my cupboards well stocked with decaffeinated tea and occasionally even decaf coffee and diet coke! I was therefore quiet a bit alarmed when I heard that the process of decaffeination introduced a slew of unwelcome chemicals into my teapot.
So, is Decaf Safe to Drink?
I’m going to lead you through all the different methods of decaffeination – sometimes it gets a bit technical as there’s a slew of information out there. This is more like the definitive guide to decaffeination rather than a simple regurgitation of the Wikipedia entry, but I’ve highlighted the main issues in each paragraph so that you can just skim through if you’re not all that particular about your decaf beverage. Let’s start with the different ways that beverages have been or are decaffeinated.
- Benzene: Decaffeination has been around for a long time. Ludwig Roselius and Karl Wimmer discovered a method involving benzene back in 1903, which marked the first commercially successful
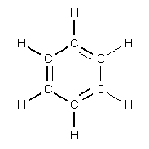 decaffeination process. There was a problem with benzene, however. Although benzene is a highly useful chemical commonly found in crude oil, it has been directly linked to cancer as well as other illnesses such as leukemia and other bone marrow problems. Scared? Yeah, you should be! Thankfully, benzene is no longer used to remove caffeine. Also banned is trichloroethylene, another organic solvent that had been used in decaffeination.
decaffeination process. There was a problem with benzene, however. Although benzene is a highly useful chemical commonly found in crude oil, it has been directly linked to cancer as well as other illnesses such as leukemia and other bone marrow problems. Scared? Yeah, you should be! Thankfully, benzene is no longer used to remove caffeine. Also banned is trichloroethylene, another organic solvent that had been used in decaffeination.
- Methylene chloride: We’ve now jumped to the present and a commonly used solvent known as methylene chloride or dichloromethane, which is colorless and possesses a sweet aroma. During the decaffeination process, the coffee beans or tea leaves are first steamed to draw the caffeine in them to the surface. Methylene chloride is then used to wash the caffeine from the beans or leaves. This process works because caffeine, but not the coffee or tea flavor, dissolves into the chemical. Although most of the chemical is removed by evaporating it off, minute traces of methylene chloride is left on the coffee or tea (I’ve seen unconfirmed sources of 1 part in a million thrown about recently. Both the US and the UK only permit a maximum of 5 parts per million). Should we be worried about 1 part in a million? Methylene chloride is common in many households products, ranging from paint stripper to pesticides. As a result, we are often exposed to it (11 parts per billion parts of air in some urban areas and waste sites according to the US Dept of Health and Human Services’ 2000 toxicology profile for methylene chloride).
So is it safe? Yes, unless you somehow drink 10,000 cups of coffee per day. High concentrations of methylene chloride can cause nausea, numbness of extremities, and even unconsciousness and death, but when I say “high concentrations,” I mean seriously high concentrations! The toxicology profile indicates that levels over 10,000 parts per million in one sitting – that’s around 10,000 cups of coffee or tea in one day – have caused death and illnesses in some animals. A study in the Journal of the American Medical Association found: “Studies of rats fed regular and decaffeinated coffee (at doses equivalent to 70 or 80 cups of coffee a day) or fed methylene chlorine in their drinking water (at doses equivalent to 125,000 to 6,250,000 cups of decaffeinated coffee a day) showed no evidence of carcinogenicity.” Convinced yet? I think like many consumers, the mere mention of chemicals even if just trace amounts may make your heart start racing. That’s probably why the imported decaf tea I drink, Typhoo (a British
 brand), doesn’t mention methylene chloride anywhere on the packaging or on their website – instead, they call it “gently” taking out the caffeine (see photo) and “a technique to wash the tea” (see their website). (Incidentally, Tetley and PG Tips both also use this same process of decaffeination). If you’re not British, then you’re probably more worried about coffee. Many brands of decaf coffee will be made using the methylene chloride method, and there’s no way to know except to check each brand (if you’re that paranoid despite what I’ve just told you about it being completely safe!). In case you are paranoid, Starbuck’s website lists Decaf Sumatra as being made using the carbon dioxide process discussed below, which doesn’t use any “chemicals” unless you’re also paranoid about CO2. I haven’t seen any definitive answer regarding other brands of Starbucks decaf although this website claims that as of 2009 all of Starbuck’s decafs were processed using methylene chloride.
brand), doesn’t mention methylene chloride anywhere on the packaging or on their website – instead, they call it “gently” taking out the caffeine (see photo) and “a technique to wash the tea” (see their website). (Incidentally, Tetley and PG Tips both also use this same process of decaffeination). If you’re not British, then you’re probably more worried about coffee. Many brands of decaf coffee will be made using the methylene chloride method, and there’s no way to know except to check each brand (if you’re that paranoid despite what I’ve just told you about it being completely safe!). In case you are paranoid, Starbuck’s website lists Decaf Sumatra as being made using the carbon dioxide process discussed below, which doesn’t use any “chemicals” unless you’re also paranoid about CO2. I haven’t seen any definitive answer regarding other brands of Starbucks decaf although this website claims that as of 2009 all of Starbuck’s decafs were processed using methylene chloride. - Ethyl Acetate or “Natural” Decaffeination: The word “natural” probably piqued your interest, although placed alongside “ethyl acetate,” it probably confused you a bit. Well, it’s “natural” because ethyl acetate occurs naturally in fruits. The process itself is similar to that explained above for methyl chloride (i.e., ethyl acetate is just used to wash the caffeine away). I haven’t found any coffee or tea brands that admit to using this process, but that may be because ethyl acetate not only takes out most of the antioxidants along with the caffeine but also imparts a chemical taste to the coffee/tea.
Are there any non-chemical methods of decaffeination? If you’re paranoid, then you’ll be happy to know that there are several non-chemical processes that a lot of companies are using these days.
- Carbon dioxide process: Pre-steamed tea leaves or coffee beans are soaked for 10 hours in a semi-liquid form of CO2 in a pressurized chamber. (CAUTION: this gets scientific here…) The CO2 binds to the caffeine molecules but leaves the flavor molecules mostly intact (although 8% of antioxidants may be removed). Then after the soaking is complete, the pressure is reduced and the CO2 evaporates and is removed along with the caffeine. You’re probably jumping for joy right now – why don’t we use CO2 to decaffeinate all teas and coffees? Well, how much do you want to pay for your precious cup of tea or coffee? Unfortunately, the high pressure required to keep the CO2 in a semi-liquid form for 10 hours does not come with a low price tag. But if you’re willing to pay for peace of mind, then go ahead. Most manufacturers that use the process happily advertise the fact. For example, I also sometimes drink Allegro’s decaf black tea and that’s decaffeinated using this process (see photo).
- Swiss Water Process of Decaffeination: You can decaffeinate with water?? Again, why doesn’t everyone just use this process?? Well, like the CO2 method, it’s also not that common and fairly expensive. Exposure of coffee beans to water multiple times may cause diminished flavor. This process is generally also only used for coffee decaffeination and not for tea. The process is not as simple as you might think and involves several stages of caffeine removal. I’ll try not to bore you with the science, but the basic process is placing the coffee beans in a caffeine-free coffee extract water. This then allows the caffeine to diffuse from the coffee beans into the caffeine-free liquid while keeping the coffee flavors in the beans because there’s already so much coffee flavors in the water. This is repeated over and over again to remove most of the caffeine. If you want a more in-depth yet understandable explanation, try this website.
Can’t I just brew the caffeine out of tea at home? I’ve read numerous sites claiming that you can decaffeinate tea yourself by throwing away the first brew of the tea. Of course, this isn’t really possible with many weak tea brands (I need to use 2 Lipton tea bags just to brew one small cup of barely drinkable tea!). While many websites suggest that it takes a mere few seconds for the caffeine to seep out of the tea, there is a very authoritative article citing studies suggesting that it will take 15 minutes of brewing before close to 100% of the caffeine is removed (30 seconds will remove only 10-20% of the caffeine). So while you can make your own cuppa decaf tea, you will also end up drinking some serious weak crap – you might as well drink hot water! It’s also possible (to a limited degree) to make a decaf your own coffee at home using the Swiss water process, but it’s rather complicated (see this website if you’re interested).
Hey, what happened to decaf soft drinks? Oh yeah, I nearly forgot about that! Did you forget what a can of coke contains? Chemicals… and you’re asking whether the chemicals in the decaffeination process is going to kill you? Well, many soft drinks actually add in caffeine extracted from the decaffeination process so I suspect that the decaf variety of those soft drinks just omit those additions.
I got lost – what’s the conclusion? Yeah, it’s taken me a few days to go through the mountainous amounts of information out there. There’s a lot of negative press about decaf drinks stemming mainly from the previous use of hazardous chemicals such as benzene. However, all the decaffeination processes today are very safe. But we all have our own individual views about what constitutes safe and the beverages industry has reacted to consumer demands for non-chemical methods of decaffeination.
Bottom line – all decaf drinks are safe, but if you jump at the word “chemical” then buy coffee or tea that has been decaffeinated using the CO2 or the Swiss water process.

